Virtual Press Conference: COVID-19 Vaccines for Children 5-11 Years Old – 10/29/2021
November 2, 2021 3:43 pm
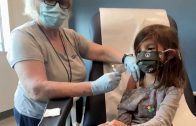
Join us for a virtual press conference to discuss the FDA’s emergency use authorization of the Pfizer-BioNTech COVID-19 Vaccine for children 5 through 11 years of age. Acting FDA Commissioner Dr. Janet Woodcock and FDA’s Center for Biologics Evaluation and Research Director Dr. Peter Marks will be taking questions.
Category: News